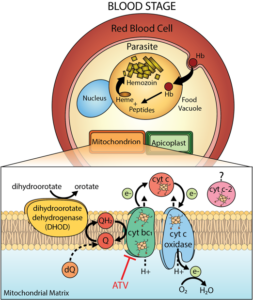
Congratulations to graduate student Tanya Espino-Sanchez and the lab of Dr. Paul Sigala in the Department of Biochemistry at the University of Utah School of Medicine for their recent publication in PNAS!! “Direct tests of cytochrome c and c1 functions in the electron transport chain of malaria parasites”. Proc Natl Acad Sci U S A 2023 May 9;120(19):e2301047120. doi: 10.1073/pnas.2301047120. Epub 2023 May 1.PMID: 37126705 PMCID: PMC10175771
Direct tests of cytochrome c and c1 functions in the electron transport chain of malaria parasites

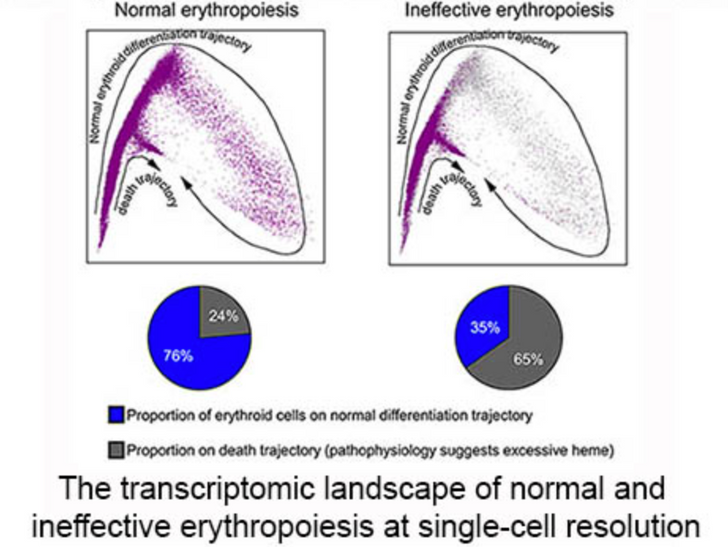
Differences in Antioxidant and Lipid Handling Protein Expression Influence How Cells Expressing Distinct Mutant TP53 Subtypes Maintain Iron Homeostasis
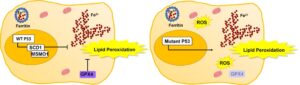

Congratulations to student Cameron Cardona and the lab of Dr. McKale Montgomery in
the Department of Nutritional Sciences at Oklahoma State University in Stillwater for
their recent publication in Cells!! “Differences in Antioxidant and Lipid Handling Protein Expression Influence How Cells Expressing Distinct Mutant TP53 Subtypes Maintain Iron Homeostasis”. Cells. 2022 Jul; 11(13): 2064. PMC9265551
Loss of the mitochondrial protein Abcb10 results in altered arginine metabolism in MEL and K562 cells and nutrient stress signaling through ATF4.
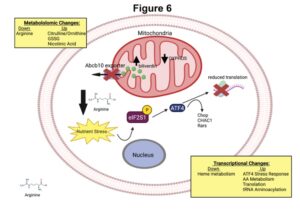

Congratulations to past Medical Laboratory Science master’s student Marisa Miljkovic and
Diane Ward, James Cox and John Phillips, members of the Utah U54 Center for Iron and Heme Disorders, for their recent JBC publication entitled “Loss of the mitochondrial protein Abcb10 results in altered arginine metabolism in MEL and K562 cells and nutrient stress signaling through ATF4“. J Biol Chem 2023 Jun 1;104877. PMDI: 37269954
This work was supported by a University of Utah CIHD P&F award DK110858 to DMW and the Mutation Generation Detection, Metabolomics and Iron and Heme Cores U54DK110858 at the University of Utah School of Medicine.
Macrophage metabolic rewiring improves heme-suppressed efferocytosis and tissue damage in sickle cell disease.
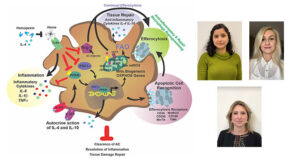
Richa Sharma, Ada Antypiuk, S. Zebulon Vance, Deepa Manwani, Quentinn Pearce, James Eric Cox, Xiuli An, Karina Yazdanbakhsh, Francesca Vinchi.
Blood 2023 Mar 23;blood.2022018026. doi: 10.1182/blood.2022018026. Online ahead of print. PMID: 36952641
This work was supported by University of Utah Center of Excellence in Hematology (CIHD) U54 DK110858 Pilot and Feasibility Grant to F.V. and the Metabolomics Core at the University of Utah School of Medicine.
Protein-metabolite interactomics of carbohydrate metabolism reveal regulation of lactate dehydrogenase.
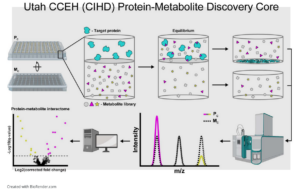

Congratulations to Kevin Hicks, James Cox and Jared Rutter, members of the Utah U54 Center for Iron and Heme Disorders, for their recent Science publication entitled “Protein-metabolite interactomics of carbohydrate metabolism reveal regulation of lactate dehydrogenase.” Science 9 Mar 2023 Vol 379, Issue 6636 pp. 996-1003.
Dr. Hicks runs the CCEH-sponsored MIDAS Core at the University of Utah School of Medicine. Check out this cutting edge core!
Mitochondrial contact site and cristae organizing system (MICOS) machinery supports heme biosynthesis by enabling optimal performance of ferrochelatase
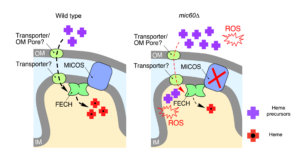

Congratulations to University of Nebraska Biochemistry Department graduate student Jonathan Dietz, from Oleh Khalimonchuk’s lab, on his paper in Redox Biol on “Mitochondrial contact site and cristae organizing system (MICOS) machinery supports heme biosynthesis by enabling optimal performance of ferrochelatase”. This study was supported by a P&F award U54DK110858.
Lysosomal iron recycling in mouse macrophages is dependent upon both LcytB and Steap3 reductases
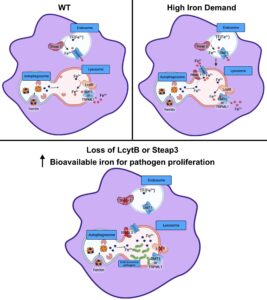

Congratulations to University of Utah Medical Laboratory Science graduate student Fanjing Meng, from Diane Ward’s lab, on her paper in Blood Advances on “Lysosomal iron recycling in mouse macrophages is dependent upon both LcytB and Steap3 reductases”. This study was supported by a P&F awarded to DMW U54DK110858.
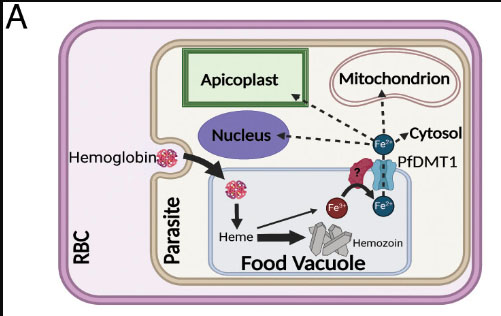
Critical role for isoprenoids in apicoplast biogenesis by malaria parasites
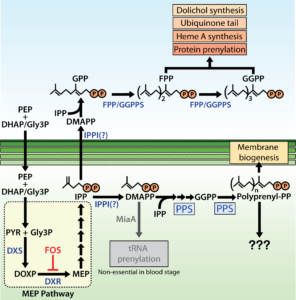

Congratulations to University of Utah Biochemistry Department graduate student Megan Okada, from Paul Sigala’s lab, on her paper in Elife on “Critical role for isoprenoids in apicoplast biogenesis by malaria parasites”. This study was supported by work from the Metabolomics Core U54DK110858.
MRP5 and MRP9 play a concerted role in male reproduction and mitochondrial function
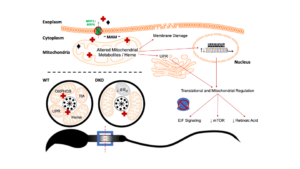

Congratulations to University of Maryland, College Park Department of Animal and Avian Sciences and Department of Cell Biology and Molecular Genetics graduate student Ian Chambers, from Iqbal Hamza’s lab, on his paper in PNAS on “MRP5 and MRP9 play a concerted role in male reproduction and mitochondrial function”. This study was supported by work from the Metabolomics Core U54DK110858.
Erythroid overproduction of erythroferrone causes iron overload and developmental abnormalities in mice
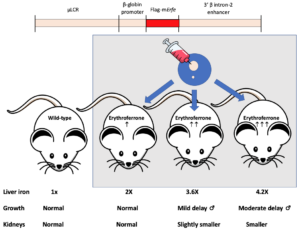

Congratulations to postdoctoral fellow Richard Coffey in the Ganz and Nemeth lab from the UCLA Center for Iron Disorders for his recent work on erythroid ERFE production and iron overload. Drs. Ganz and Nemeth are members of the Utah CCEH.
The biochemical basis of mitochondrial dysfunction in Zellweger Spectrum Disorder
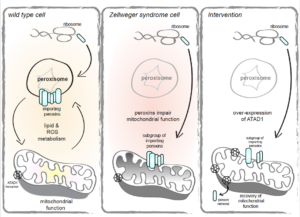

Congratulations to postdoctoral fellow Esther Nuebel from Jared Rutter’s lab in the Department of Biochemistry at the University of Utah School of Medicine for her recent work on Zellweger Spectrum Disorders and mitochondrial dysfunction. Dr. Rutter is a member of the Utah CIHD and the Metabolomics/Lipidomics core, which is funded by the U54 CIHD, was utilized in this study.
Divergent acyl carrier protein decouples mitochondrial Fe-S cluster biogenesis from fatty acid synthesis in malaria parasites
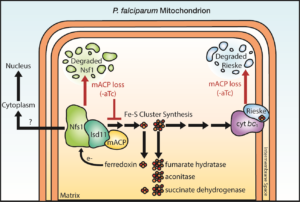

Congratulations to University of Utah Biochemistry Department graduate student Seyi Falekun, from Paul Sigala’s lab, on his paper in Elife on “Divergent acyl carrier protein decouples mitochondrial Fe-S cluster biogenesis from fatty acid synthesis in malaria parasites. This work was initially funded by a CIHD pilot award.
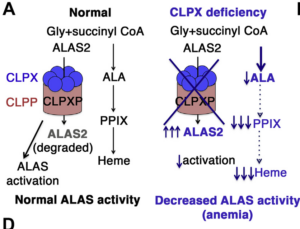
The ubiquitous mitochondrial protein unfoldase CLPX regulates erythroid heme synthesis by control of iron utilization and heme synthesis enzyme activation and turnover
Rondelli CM, Perfetto M, Danoff A, Bergonia H, Gillis S, O’Neill L, Jackson L, Nicolas G, Puy H, West R, Phillips JD, Yien YY. J Biol Chem. 2021 Jul 16:100972. doi: 10.1016/j.jbc.2021.100972. Online ahead of print. PMID: 34280433
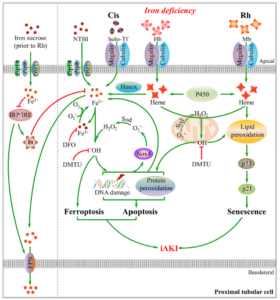
Iron deficiency exacerbates cisplatin- or rhabdomyolysis-induced acute kidney injury through promoting iron-catalyzed oxidative damage
Zhao S, Wang X, Zheng X, Liang X, Wang Z, Zhang J, Zhao X, Zhuang S, Pan Q, Sun F, Shang W, Barasch J, Qiu A. Free Radic Biol Med. 2021 Jul 21;173:81-96. doi: 10.1016/j.freeradbiomed.2021.07.025. Online ahead of print. PMID: 34298093
Iron-dependent apoptosis causes embryotoxicity in inflamed and obese pregnancy
Fisher AL, Sangkhae V, Balušíková K, Palaskas NJ, Ganz T, Nemeth E. Nat Commun. 2021 Jun 29;12(1):4026. doi: 10.1038/s41467-021-24333-z. PMID: 34188052